Sex differences in the association of wearable accelerometer-derived physical activity with coronary heart disease incidence and mortality
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) (Supplementary Table 15). All data field identifiers are listed in Supplementary Table 16.
Study population
The UK Biobank is extensively described online (www.ukbiobank.ac.uk). Briefly, over 500,000 participants aged 37–73 years were recruited between 2006 and 2010 from 22 assessment centers across England, Scotland and Wales. Each participant completed a touch-screen questionnaire, underwent a nurse-led interview, had physical measurements taken and provided biological samples27. This study was conducted under UK Biobank application number 134551. UK Biobank had ethical approval from the North West Multicenter Research Ethics Committee (ref: 11/NW/0382). All participants gave written informed consent.
Accelerometer-derived PA
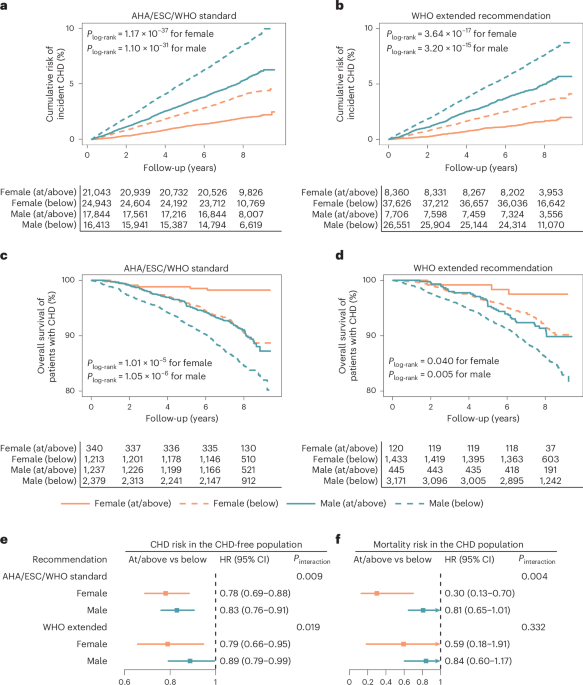
In the PA sub-study conducted between February 2013 and December 2015, 103,695 participants provided the weekly wrist-worn triaxial accelerometer (Axivity AX3) data28,29. We excluded individuals who withdrew consent and had unreliable data size (field 90002), insufficient wear time (field 90015), poor calibration (field 90016, 90017) and implausible acceleration values (field 90012). We further excluded individuals with missing covariates or who developed CHD during the wearing period. Finally, 85,412 eligible participants were retained, among which 80,243 without CHD were included in the CHD incidence study to analyze the CHD incidence risk, and 5,169 with established CHD were included in the CHD mortality study for the mortality risk of CHD patients; see details in Supplementary Figs. 10 and 11.
As described previously, PA intensity was quantified as the average vector magnitude over 5-s epochs recorded by a wrist-worn triaxial accelerometer, expressed in milligravity (mg). Non-wear-time epochs were identified as consecutive stationary episodes ≥60 min in which all three axes had standard deviation <13.0 mg, which were imputed on the basis of the average of similar time-of-day vector magnitude and intensity distribution data points on different days30. Furthermore, MVPA duration was defined as the accumulation of 5-s epochs with mean acceleration ≥100 mg (refs. 30,31,32). To minimize the artifact misclassification of MVPA, we extracted MVPA data in bouts defined as 5-min periods in which more than 80% of epochs met the intensity threshold30,31,32. Additional measures derived from the quantified MVPA duration were as follows: (1) adherence to the standard recommendations of the AHA2, ESC3 and WHO4 (AHA/ESC/WHO standard: ≥150 min week−1); (2) adherence to the extended recommendation of WHO (WHO extended: ≥300 min week−1); (3) number of days the average daily AHA/ESC/WHO standard (≥150/7 min d−1) was met and (4) number of days the average daily WHO extended recommendation (≥300/7 min d−1) was met.
Outcome ascertainment
For the CHD incidence study, the time-to-event outcome was defined as following-up years from the end of wear time to incident CHD in the CHD-free population, ascertained by the first occurrences of any code mapped to three-character International Classification of Diseases 10th Revision codes I20–I25 from primary care, hospital records, death registries and self-report fields. For the CHD mortality study, the outcome was following-up years from the end of wear time to the date of all-cause death in the established CHD population. Censoring was defined as death, withdrawal, loss to follow-up or the end of the follow-up period, whichever occurred first.
Covariates
Demographics including age, gender, center (England, Scotland or Wales), ethnicity (white or other), education (college, university or other), Townsend deprivation index and body mass index (BMI); lifestyles including smoking (never, ever or current), alcohol (never, ever or current), sleep duration (<7 h, 7–8 h or >8 h) and dietary health33,34 (Supplementary Table 17); medical conditions including diabetes, hypertension, dyslipidemia, cholesterol-lowering medication, blood pressure medication, insulin therapy and Charlson comorbidity index35; and activity intensity measured by average acceleration were included as common covariates in the CHD incidence and mortality study. We further adjusted the polygenetic risk score of CHD in the CHD incidence study, and the use of CHD treatment medicines (antiplatelet medication, antianginal medication) in the CHD mortality study.
Cox proportional hazard models
Models with follow-up time as the timescale
For participant i at follow-up time t (years from the end of accelerometer wear to the event of interest or censoring), the hazard function was:
$${h}_{i}(t)={h}_{0}(t)\exp (\,\beta {X}_{i}\,+\,\gamma {C}_{i})$$
where Xi represents the PA measures, Ci represents the covariates and h0(t) is the baseline hazard at time t. Sex-specific associations were estimated from separate Cox models for females (gi = 0) and males (gi = 1):
$${h}_{i}(t\,|\,{g}_{i})\,=\,{h}_{0,{g}_{i}}(t)\exp (\,{\beta }_{{g}_{i}}{X}_{i}\,+\,{\gamma }_{{{\rm{g}}}_{i}}{C}_{i})$$
where baseline hazard \({h}_{0,{g}_{i}}(t)\) varies by sex, and \({\beta }_{{g}_{i}}\) denotes the sex-specific effects.
Interactions between sex and PA measures were tested using a likelihood ratio test, comparing the following two models with or without an interaction term.
$$\begin{array}{l}{{\rm{M}}{\rm{o}}{\rm{d}}{\rm{e}}{\rm{l}}}_{1}:\,{h}_{i}(t)\,=\,{h}_{0}(t)\exp (\,{\beta }_{1}{X}_{i}\,+\,{\beta }_{2}{g}_{i}\,+\,\gamma {C}_{i}\,+\,\theta ({X}_{i}\,\times \,{g}_{i}))\\ {{\rm{M}}{\rm{o}}{\rm{d}}{\rm{e}}{\rm{l}}}_{2}:\,{h}_{i}(t)\,=\,{h}_{0}(t)\exp (\,{\beta }_{1}^{{\prime} }{X}_{i}\,+\,{\beta }_{{2}}^{{{\prime} }}{g}_{i}\,+\,{\gamma }^{{\prime} }{C}_{i})\end{array}$$
Models with age as the timescale
When age was used as the timescale, the hazard for participant i at age a was:
$${h}_{i}(a)={h}_{0}(a)\exp (\,\beta {X}_{i}+\gamma {C}_{i})$$
where \({h}_{0}(a)\) represents the baseline hazard at age a. Participants entered the risk set at their attained age and were followed until the age at the event or censoring. Sex-specific associations and sex–PA interactions were evaluated analogous to the follow-up time models.
Stratified Cox models by sex
In stratified Cox models36, the baseline hazard \({h}_{0,{g}_{i}}(t)\) was allowed to differ by sex:
$${h}_{i}(t\,|\,{g}_{i})={h}_{0,{g}_{i}}(t)\exp (\,\beta {X}_{i}+\gamma {C}_{i})$$
To test sex difference, an interaction term between sex and PA measures was included:
$${h}_{i}(t\,|\,{g}_{i})={h}_{0,{g}_{i}}(t)\exp (\,\beta {X}_{i}+\gamma {C}_{i}+\theta ({X}_{i}\times {g}_{i}))$$
For females (gi = 0), the effect of PA measures is simply β. For males (gi = 1), the effect is β + θ. The significance of the sex difference was assessed by likelihood ratio test, comparing models with or without an interaction term.
$$\begin{array}{l}{\mathrm{Model}}_{1}:\,{h}_{i}(t\,|\,{g}_{i})={h}_{0,{g}_{i}}(t)\exp (\,\beta {X}_{i}+\gamma {C}_{i}+\theta ({X}_{i}\times {g}_{i}))\\ {\mathrm{Model}}_{2}:\,{h}_{i}(t\,|\,{g}_{i})={h}_{0,{g}_{i}}(t)\exp (\,\beta ^{{\prime} }{X}_{i}+\gamma^{{\prime} } {C}_{i})\end{array}$$
Statistical analyses
Descriptive characteristics were presented as mean ± s.d. or median with interquartile range for quantitative variables as appropriate; categorized variables were described by frequency (n) and proportion (%). The cumulative risk of incident CHD across guideline-adherent groups was illustrated using a cumulative incidence curve, and the overall survival for patients with CHD across groups was shown using a Kaplan–Meier survival curve. Event proportions over the follow-up period and event rates per 100,000 person-years were calculated to describe the outcome characteristics across groups. Cox proportional hazard models with follow-up time as timescale were used to examine sex-specific associations of all PA measures with both CHD incidence and mortality. HRs and 95% CIs were estimated from models to measure relative instantaneous risk of the outcome, and we reported the HR per 30 min week−1 on MVPA duration. Nonlinear relationships and dose–response associations between MVPA duration and CHD outcomes were evaluated through restricted cubic splines by using R package ‘rms’, with 0 min week−1 of MVPA as the reference. Sex differences in associations were assessed using likelihood ratio tests, comparing models with and without interaction terms between sex and PA measures37. Given the distinct populations and different hypotheses underlying the two outcomes, we applied false discovery rate (FDR) correction using the Benjamini and Hochberg method38 for CHD incidence and mortality separately to account for multiple comparisons across five PA measures evaluated in each analysis.
We performed several sensitivity analyses. (1) We used Fine and Gray subdistribution hazard models39 to account for the competing risk of death in the CHD incidence study; (2) we additionally fitted Cox proportional hazard models with age as the timescale to further control for potential confounding by age; (3) we used stratified Cox proportional hazard models by sex to account for sex-specific baseline hazards36; (4) we examined multiple PA measures with varying thresholds; (5) we used four models with progressively increasing covariate adjustment, and further conducted sensitivity analyses incorporating additional comorbidities (chronic kidney disease and arthritis) as well as family history (biological father, mother and siblings) of heart disease, to account for potential confounders; (6) we excluded participants with events occurring within the first year of the follow-up period to mitigate reverse causality; and (7) we imputed the missing data using the multiple imputation method with the R package ‘mice’40.
Statistical analyses were conducted using R v.4.3.3, and a two-sided P ≤ 0.05 was considered statistically significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
link



:max_bytes(150000):strip_icc()/A-Physical-Therapist-Shares-the-Most-Underrated-Physical-Activity-94666d193c98412ca90ffbcc23339f90.jpg)



