Physical activity levels may impact on the risk of premature mortality in people with epilepsy
Study population
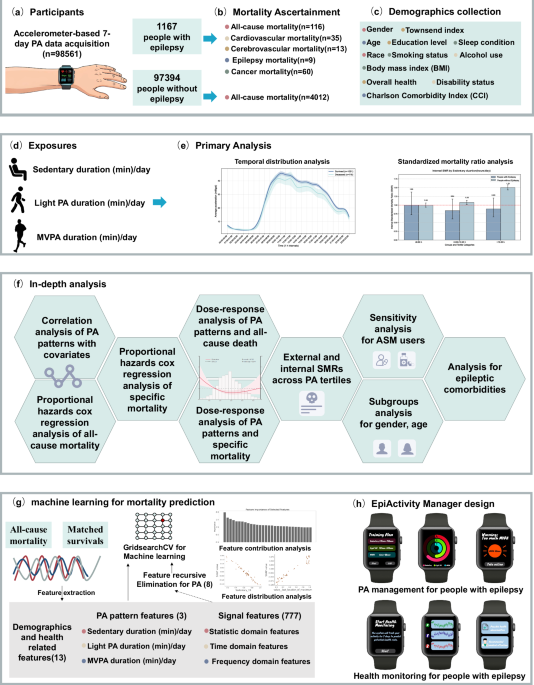
This prospective cohort study analysed data from 98,561 UK Biobank participants who provided valid wrist-worn accelerometer data, including 1167 people with epilepsy and 97,394 people without epilepsy. The UK Biobank is a comprehensive longitudinal study that recruited over 500,000 participants aged 40–69 between 2006 and 2010. The North West Multi-Centre Research Ethics Committee approved the study (R21/NW/0157), and all participants provided informed consent35. This study was conducted under UK Biobank application number 162384 (epilepsy and comorbidity), with the research protocol reviewed and approved as part of the original data access application process. Between February 2013 and December 2015, 236,519 UK Biobank participants were invited to participate in an accelerometer study. Of these, 106,053 agreed to participate and wore wrists that were worn accelerometers (Axivity AX3). The UK Biobank accelerometer expert working group processed the raw accelerometer data from 103,682 participants. For our analysis, we included participants who had at least three days (72 h) of accelerometer data and coverage of each hour of the 24-h cycle. Hourly sedentary behaviour, LPA, and MVPA metrics were derived from the raw accelerometer data (field ID: 1020:40032, 40033, 40034).
We integrated self-reported epilepsy diagnoses and relevant coding in electronic health records. According to the International Classification of Diseases 9th (ICD-9) and 10th (ICD-10) editions, epilepsy codes included ICD-9: 345 (epilepsy), ICD-10: G40 (epilepsy). Additionally, we excluded 349 persons with ICD-10 code R56 (convulsions, not elsewhere classified) from the group of people without epilepsy. The final sample included 1167 people with epilepsy and 97,394 people without.
Accelerometer-based physical activity measurement and processing
Participants were instructed to wear an Axivity AX3 triaxial accelerometer on their dominant wrist for 7 days, continuously including sleep. The device recorded acceleration at 100 Hz with a dynamic range of ± 8 g. Raw acceleration signals were aggregated into 5-second epochs, representing mean vector magnitude. Detailed data collection and processing methods are available in the provided reference36. UK Biobank researchers employed previously published machine learning algorithms to classify activity behaviours into accurately sedentary, LPA, MVPA and sleep based on wrist accelerometer data. This showed an 88% classification accuracy with a Cohen’s kappa coefficient of 0.803. The average proportion of time spent in sedentary, LPA, and MVPA was estimated for each hour during wakefulness. Data were stored as comma-separated values of 24, each representing one hour of the day, from midnight to 11 PM.
Mortality ascertainment
The primary outcome of this study was all-cause mortality among people with epilepsy. Secondary outcomes included cause-specific mortality, focusing on epilepsy-related, cardiovascular disease, cerebrovascular disease, and cancer mortality. Mortality information was obtained through the NHS Central Register death registration databases. Cause-specific mortality was determined based on the ICD-10. Death dates and causes were retrieved from NHS Central Register death datasets. Follow-up time was estimated from the date of completing accelerometer wear until death, withdrawal from the study, or the last follow-up date (October 17, 2023), whichever occurred first.
Covariate assessment
We included the following covariates: demographic factors (age, gender, ethnicity, education level, Townsend deprivation index), lifestyle factors (smoking status, alcohol consumption), health status indicators (disability status, overall health rating), and comorbidities index. Age was measured in years as a continuous variable. Ethnicity was classified as white or others. Education level was categorised into two groups: those with a bachelor’s degree or above, and those without a degree. The Townsend deprivation index, a measure of material deprivation based on unemployment, non-car ownership, non-home ownership, and household overcrowding, was calculated from participants’ postcodes at recruitment, with higher scores indicating greater deprivation. Smoking status was categorised as never, previous, or current smoker. Alcohol consumption was similarly classified as never, previous, or current. Disability status was self-reported as either present or absent. Overall health rating was self-reported on a four-point scale: excellent, good, fair, or poor. Comorbidity index was obtained through self-reported medical history and hospital records. We estimated a Charlson Comorbidity Index (CCI) based on the UK Biobank ICD-10 records to quantify the comorbidity burden. The CCI assigns weights to various comorbid conditions (i.e., myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, connective tissue disease, peptic ulcer disease, mild liver disease, diabetes without chronic complication, diabetes with chronic complication, hemiplegia or paraplegia, renal disease, any malignancy (including leukemia and lymphoma), moderate or severe liver disease, metastatic solid tumor, and AIDS/HIV) and sums these weights to create a composite score that predicts mortality risk.
Statistical analysis
Statistical analysis incorporated three main components: exploratory data analysis, survival analysis and nonlinear relationship exploration, subgroup and sensitivity analysis and machine learning-based mortality prediction. We used Python 3.11 and R 4.4.1 for the analysis. We applied Benjamini-Hochberg to perform multiple comparisons when assessing the three PA variables (sedentary behaviour, LPA, and MVPA) in primary analysis and subgroup analysis.
We conducted exploratory data analysis, describing continuous variables using means and standard deviations, and categorical variables as frequencies and percentages. Differences between groups were assessed using t-tests for continuous variables and χ² tests for categorical variables. Missing data were imputed using the MISSFOREST algorithm37.
We assessed relationships between the three exposure variables (sedentary behaviour, LPA and MVPA) and covariates using Spearman correlation analysis. We further examined the strength and direction of associations for continuous variables, which showed significant correlations. For categorical variables, we investigated differences in physical activity patterns across different categories using t-tests. To ensure our analyses were not affected by multicollinearity, we calculated variance inflation factors (VIF) among sedentary behaviour, LPA, and MVPA. The VIF values ranged from 1.05 to 1.96, indicating low multicollinearity among these variables (Supplementary Table 17).
We conducted SMR analyses to account for differences in follow-up duration, age, and gender distribution. The SMR was estimated as the ratio of observed deaths to expected deaths. For the epilepsy group, expected deaths were estimated based on age-specific (5-year bands) and gender-specific mortality rates from the people without epilepsy, which served as the reference population.
We also examined SMRs across tertiles of sedentary behaviour, LPA, and MVPA to explore potential nonlinear relationships. We estimated external SMRs (comparing each tertile in the epilepsy group to the corresponding tertile in the group of people without epilepsy) and internal SMRs (using the first tertile within each group as the reference). These analyses provided insights into how mortality risk in epilepsy varies across different levels of physical activity while accounting for demographic differences.
We began our survival analysis by examining the interaction effects between PA patterns and the presence or absence of epilepsy on mortality risk in the entire cohort using Cox proportional hazard models. We then employed Cox proportional hazard models to evaluate relationships between the three PA exposures (sedentary behaviour (hours/day), LPA (hours/day), and MVPA (hours/day)) and all-cause and cause-specific mortality risks, estimating hazard ratios (HRs) and their 95% confidence intervals (CIs) for people with epilepsy and people without epilepsy.
We constructed three models with increasing levels of adjustment: Model 1 included only the three PA exposure indicators (sedentary behaviour (hours/day), LPA (hours/day), and MVPA (hours/day)); Model 2 incorporated demographic variables (gender, age, BMI, ethnicity, education level and lifestyles) and socioeconomic status (Townsend Deprivation Index); Model 3 (fully adjusted model) included all variables from Model 2 and additionally adjusted for health conditions (Overall health, disability status, and comorbidity index).
To explore potential nonlinear relationships between PA patterns and mortality, we applied RCS models based on the fully adjusted Cox proportional hazards regression (Model 3). RCS models were optimised using Akaike Information Criterion to determine the optimal number of knots. This analysis allowed us to investigate dose-response relationships between continuous measures of physical activity (sedentary behaviour, LPA, and MVPA) and mortality in people with epilepsy and people without epilepsy.
Within the epilepsy group, we conducted stratified analyses by gender (men, women) and age subgroups (<65 years, ≥65 years), repeating Cox proportional hazard models and RCS analyses. We further tested for interactions effect between PA patterns and five major comorbidities (diabetes, hypertension, cardiovascular disease, cerebrovascular disease, and peripheral vascular disease). Stratified analyses were conducted only for comorbidities with significant interactions.
As a sensitivity analysis, we applied a definition of epilepsy that requires a self-report or ICD-based diagnosis and documented use of an antiseizure medication (ASM). They were systematically identified according to four major described mechanisms of action38. (1) sodium channel blockers, (2) gamma-aminobutyric acid (GABA) analogues, (3) synaptic vesicle protein 2 A (SV2A) binding agents, and (4) agents with multiple mechanisms. Using these criteria, we identified 527 individuals as ASM users.
Lastly, we developed a mortality prediction model based on machine learning for people with epilepsy. We first employed propensity score matching (PSM) based on age and gender to create balanced groups of people with epilepsy either deceased or alive. The dataset was then randomly split into a training set (80%) and a testing set (20%). We extracted features from the minute-epoch acceleration summaries using the ‘tsfresh’ library, which generated comprehensive time and frequency domain features from the raw accelerometer signals15. To improve computational efficiency and reduce overfitting, we applied random forest-based recursive feature elimination (RFE) to select the most informative features from the initial pool of 777 time-frequency domain features. We retained 8 key features for the final model on the training sets.
We developed multiple prediction models with increasing complexity: (1) a baseline random forest model including demographic factors (age, gender, ethnicity, education level, Townsend deprivation index), lifestyle factors (smoking status, alcohol consumption), health status indicators (disability status, overall health rating), comorbidities status and ASM-related variables, and (2) the full random forest model incorporating the selected PA patterns (sedentary duration, LPA duration, MVPA duration) and time-frequency features ((Mean change in moderate activity, High-frequency PA intensity (FFT 33), High-frequency PA (imaginary, FFT 16), Very high-frequency PA phase (FFT 56), Low-frequency PA (imaginary, FFT 3), Activity variability (std), Ultra high-frequency PA (imaginary, FFT 91), Short-term PA trend (5-epoch max))). Hyperparameter optimisation was performed exclusively on the training dataset using GridSearchCV (Supplementary Table 18)39.
Model performance was evaluated using accuracy, area under the receiver operating characteristic curve (AUROC), sensitivity, specificity, F1 score, and area under the precision-recall curve (AUPRC). We conducted ablation studies to quantify the incremental value of accelerometer-derived features beyond traditional clinical predictors. All model development and reporting adhered to the TRIPOD + AI guidelines for transparent reporting of prediction models using machine learning methods40.
To enhance interpretability, we analysed feature importance using Shapley Additive Explanations (SHAP) values41, which quantify each feature’s contribution to individual predictions. We also explored the relationship between the predicted probability (fully adjust-model) of death and actual time-to-death using Spearman correlation analysis and created calibration plots to visualise the agreement between predicted probabilities and observed mortality rates in the test set.
link



:max_bytes(150000):strip_icc()/A-Physical-Therapist-Shares-the-Most-Underrated-Physical-Activity-94666d193c98412ca90ffbcc23339f90.jpg)



